
FJSL
Max Gold, a 15-year-old student from St. George’s International School, is one of the winners of the 2021 National Jonk Fuerscher contest.
Max Gold, a 15-year-old student from St. George’s International School, has created a computer programme to simplify learning about properties of chemical elements and how they interact with each other. He used an existing programming platform, called Python, to write a computer code that can help students to understand and practice valency manipulations and balancing chemical reactions. As the data source for this functionality, Max has created an extensive database, which also serves as a dictionary of elements and compounds that can be used by students while they use his learning tool. Although there are online programs supporting learning about a broad range of scientific topics, Max believes that his approach is more specific and elegant and may therefore be more helpful to the users.
Max presented his project during the 2021 National Jonk Fuerscher contest, organised by Fondation Jeunes Scientifiques Luxembourg (FJSL) and was chosen as one of the winners.
What does it mean to “balance chemical reactions”?
Elements and chemical compounds are the building blocks of every material, liquid and gas that we have in our surroundings. These elements and compounds can be fused or broken down to create new compounds in a process of chemical reactions. Chemical reactions are the base of all processes in life: they allow us to breathe, to turn food into energy and to drive a car. According to the principle of conservation of mass, the number of elements before the reaction must be equal to the number of elements resulting from the reaction. Therefore, balancing a chemical reaction means that an equation representing that reaction must contain the correct numbers of all elements and compounds in the correct combinations. Chemical reactions fall into one of the four basic types:

What is the valency of elements?
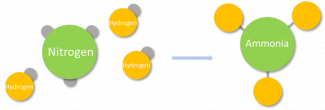
Valency is the measure of the willingness of one element to react with another element or compound. It is determined by the number of small particles, electrons, that are available to form a chemical bond with other elements. This number determines how willing the element is to react and how many other elements it can react with. Two elements or compounds always have a tendency to create the simplest possible compound between them. For example, nitrogen has a valency of 3 and hydrogen has a valency of 1. Together they will form ammonia, or NH3, which has 3 bonds:
How did Max get the idea to write this program?
Max has loved numbers for as long as he can remember. One of his first memories involves pre-school games recognising and organizing patterns of numbers, which made him really happy. Ever since, mathematics has been Max’s favourite subject at school.
While Max was learning programming during a Computer Sciences course in high school, he wondered whether he could automate the way he was learning chemistry, so that he and students like him could prepare for exams in a simpler, more efficient way. The first topic he focussed on was calculating the valency of elements and chemical compounds. As Max remembers: “the code was not written very well, but I managed to make it work. I could check my answers while studying chemistry and it was awesome!” Encouraged by the success of this module, and following discussions with his chemistry and maths teachers, Max moved onto a module focussing on balancing chemical reactions. This was much more challenging, as the underlying maths was more difficult, but the Covid-related lockdown provided the opportunity to work on this until completion.
How did Max experience the Jonk Fuerscher contest?
Max was the first participant of Jonk Fuerscher contest from his high school. Even though he was nervous about presenting his work in front of such a large audience, he really enjoyed this experience and is very thankful for it. One of the highlights for Max was the presence of a very special guest in the audience: “Only after 15 minutes of presenting, have I realized that I have been, in fact, presenting my work to the Prince of Luxembourg. And I had no idea!” HRH Guillaume, the Hereditary Grand Duke of Luxembourg, attended this year’s national contest.
What are Max’ plans for the future?
As a reward for winning the Jonk Fuerscher contest, Max will participate in the Taiwan International Science Fair in February 2022. Before this time, he is planning to create a website for everybody to have access to his easy-to-use learning tool. He also hopes to include two new modules on topics from advanced maths on his website.
After finishing high school Max is planning to study mathematics.
Author: Anna Keller
Editor: Michèle Weber (FNR)






